[UNKNOWN ORGANIC REACTIONS] Unknown Mechanisms & Novel Reactions JC CHEM SITCOMS
Video Preview Thumbnail.
1 / 15
2 /
15

3 /
15
4 /
15
5 /
15
6 /
15
7 /
15

8 /
15

9 /
15

10 /
15

11 /
15

12 /
15
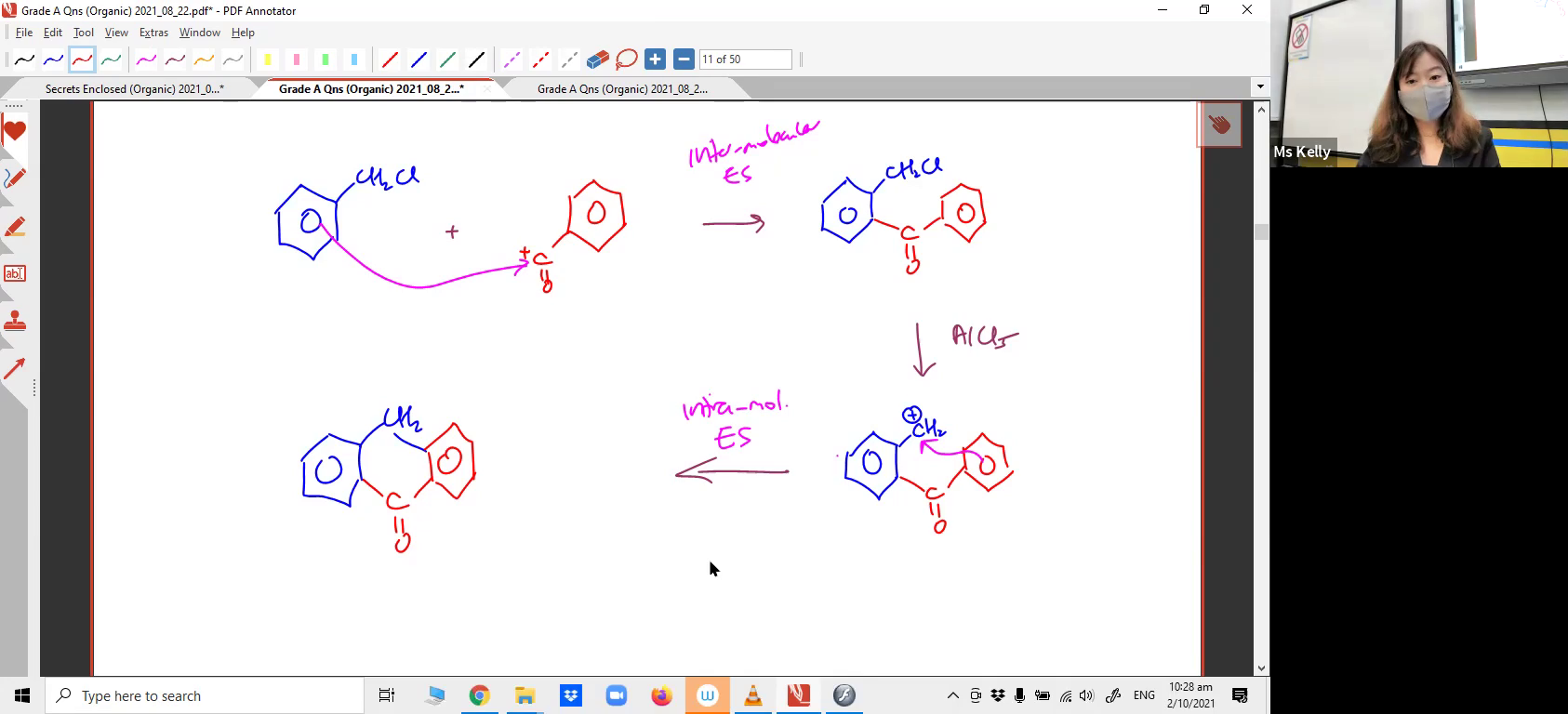
13 /
15

14 /
15
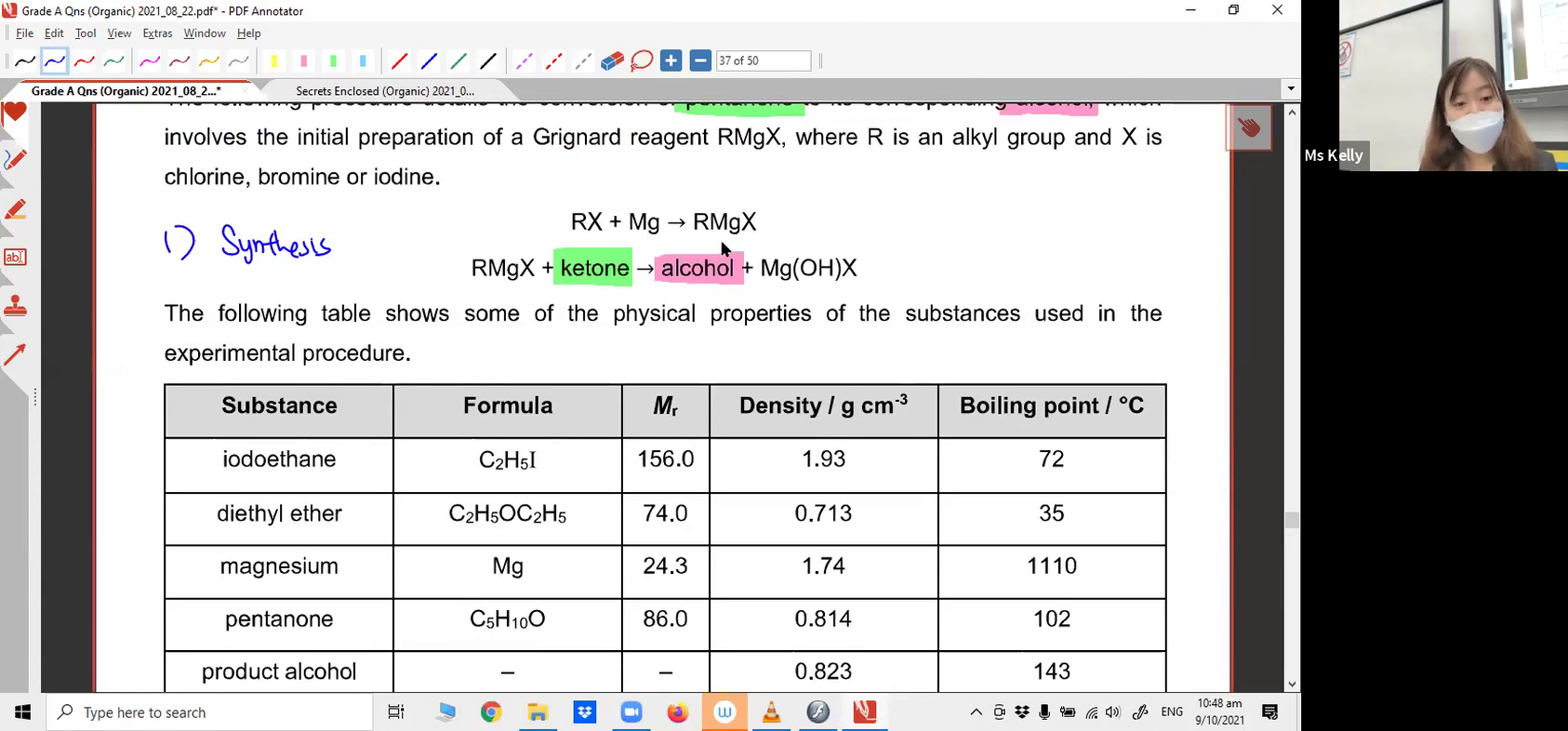
15 /
15















Overview
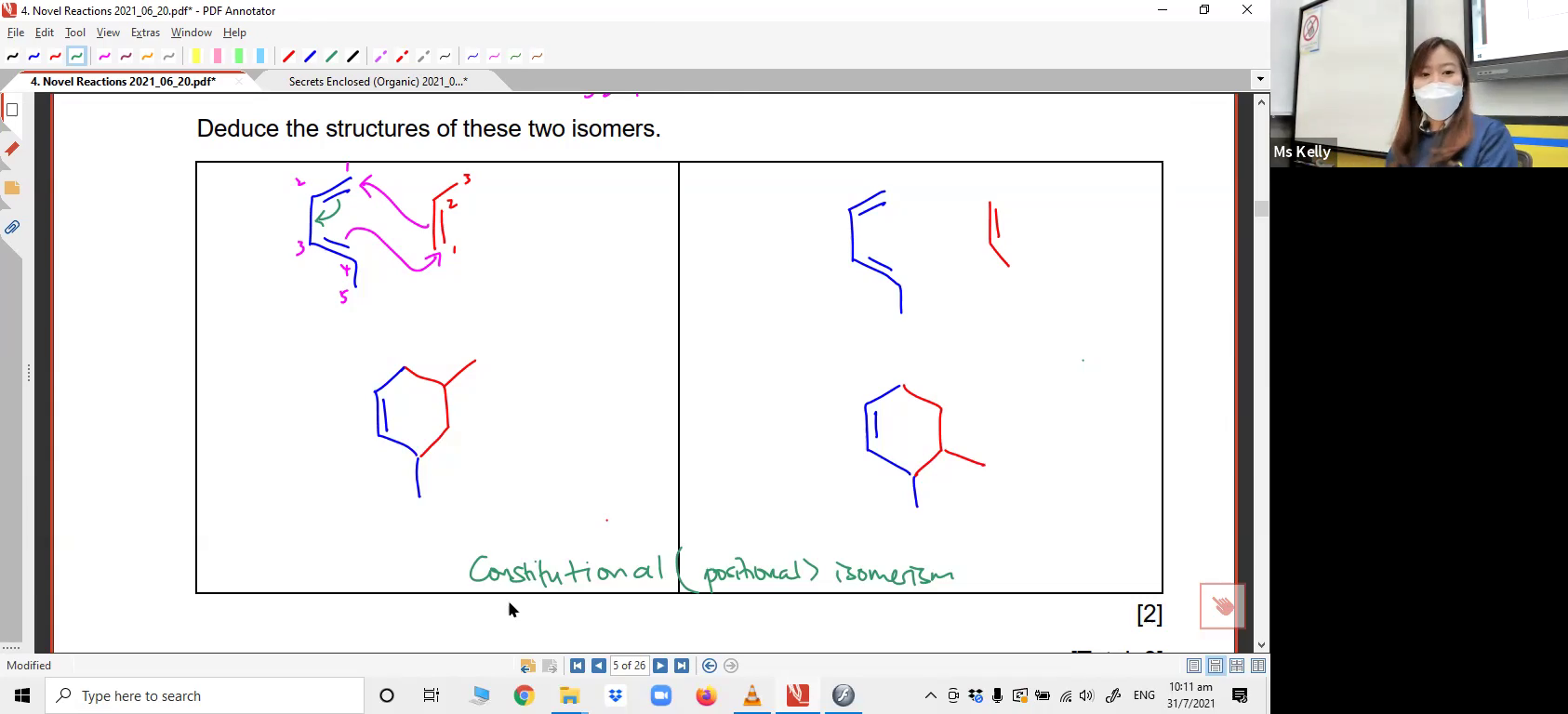
1. Diels-Alder Reaction
A compilation of data-based questions that can appear in Paper 1, 2 or 3! Acquire observation and deduction skills that'll allow you to tackle any kind of reactions and mechanisms you've never seen before! (Diels-Alder Reaction) Applying Diels-Alder reaction to generate cyclic compounds via pi electron pair movement.2. Methyl Shift
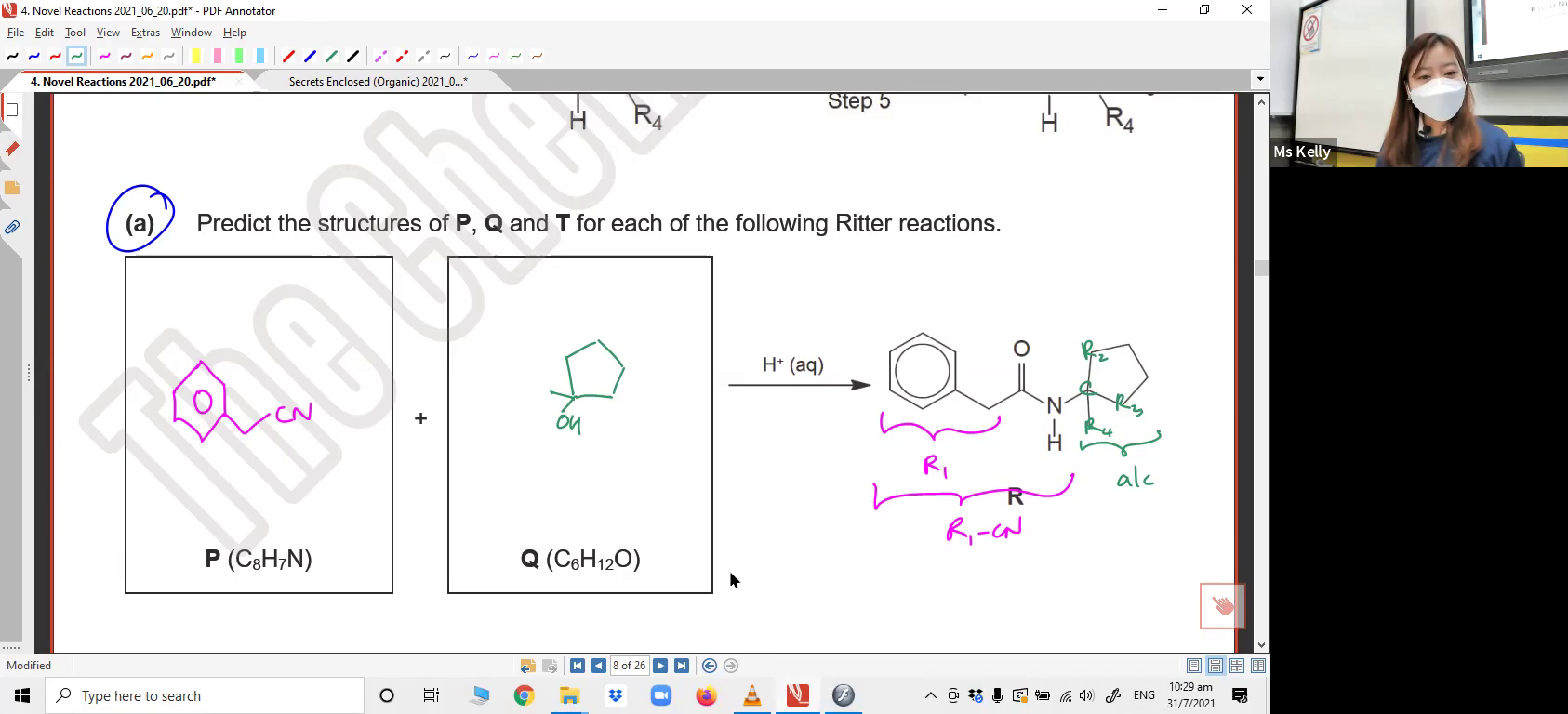
Understanding methyl shift to form more stable carbocation. Illustrating the mechanism. Suggesting product formed from applying methyl shift.3. Ritter Reaction
Understanding the pattern of Ritter reaction to form amide from nitrile and tertiary alcohol. Applying the pattern to suggest reactants and products. Illustrating the mechanism. Explaining the slow step of the mechanism.4. Formation of ethers from alcohols
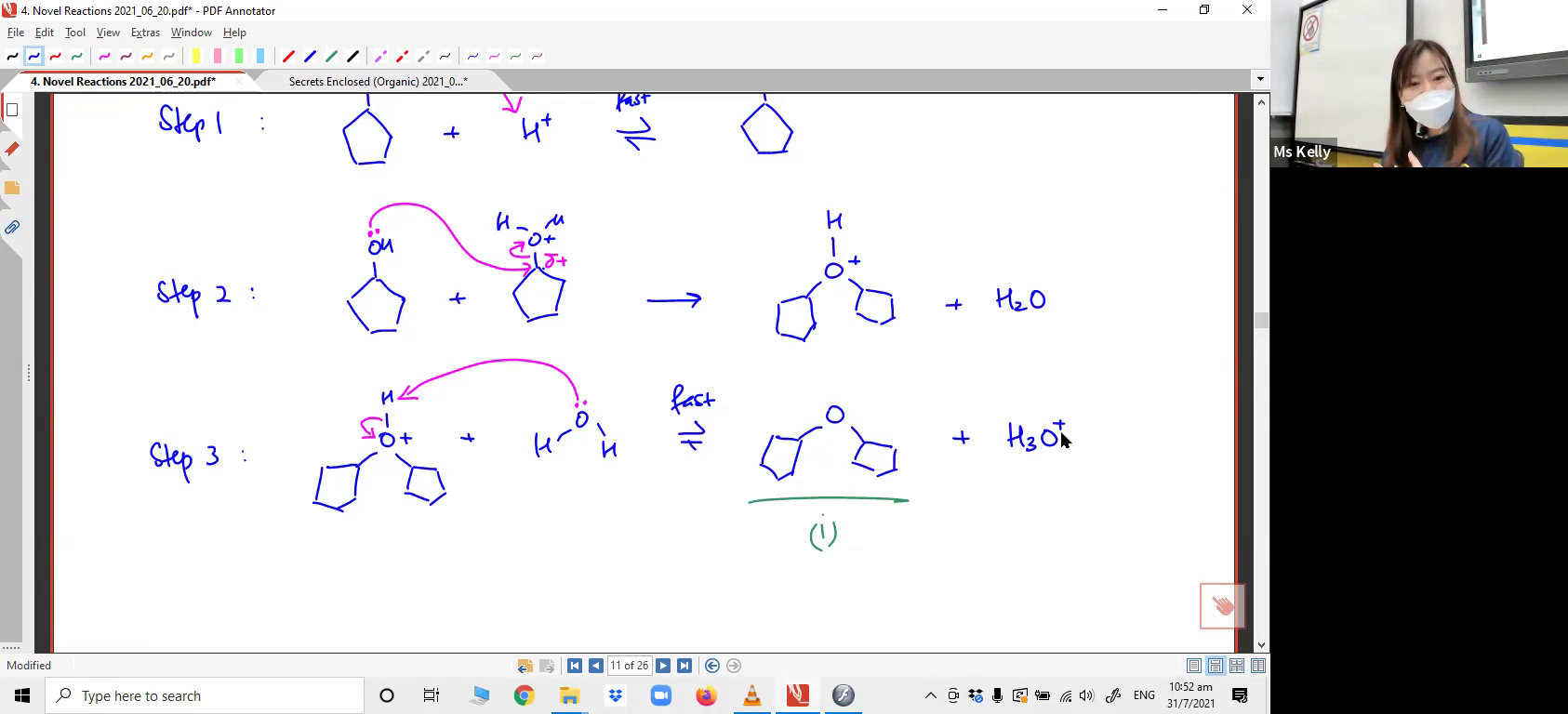
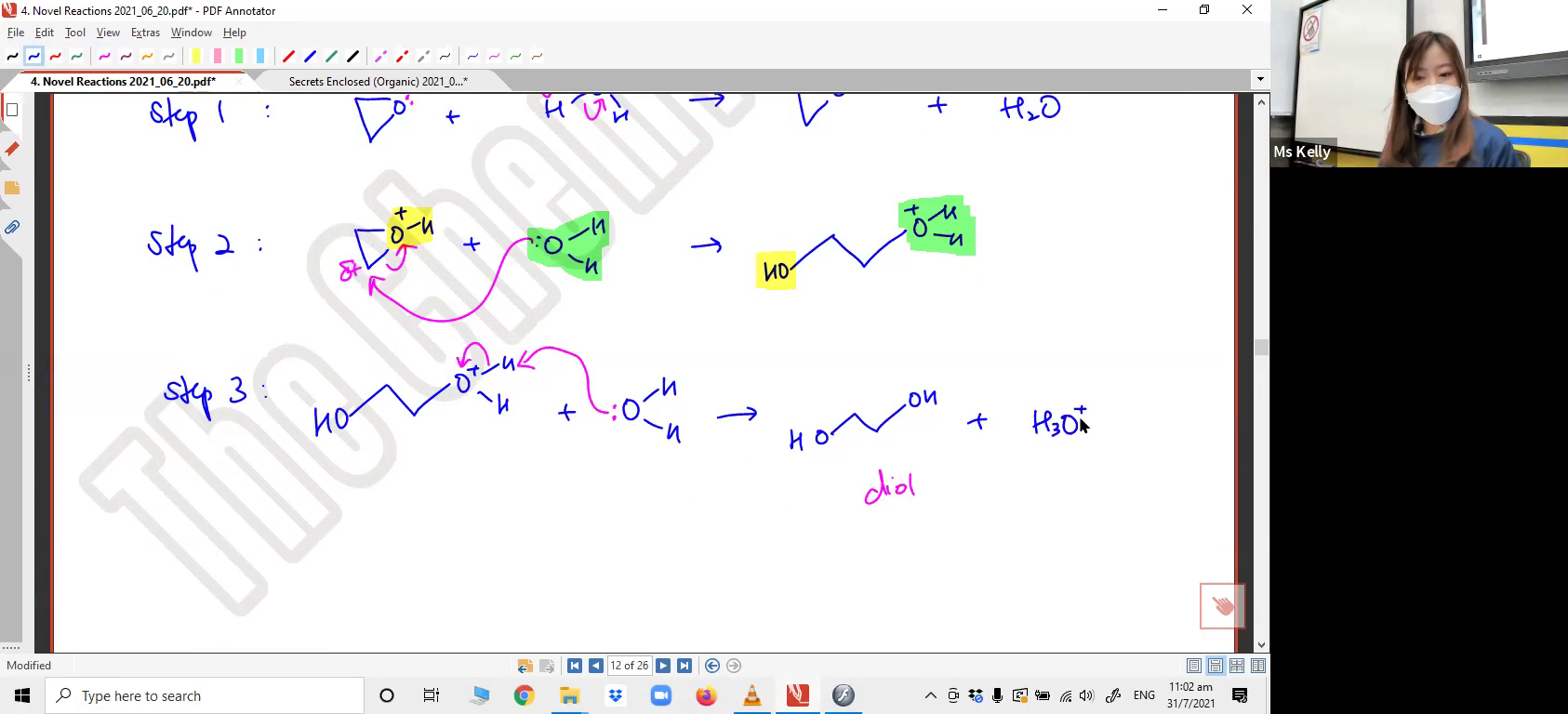
Drawing the full mechanism for the reaction between two cyclopentanol molecules to form an ether.5. Reaction involving Epoxides, cyclic ethers
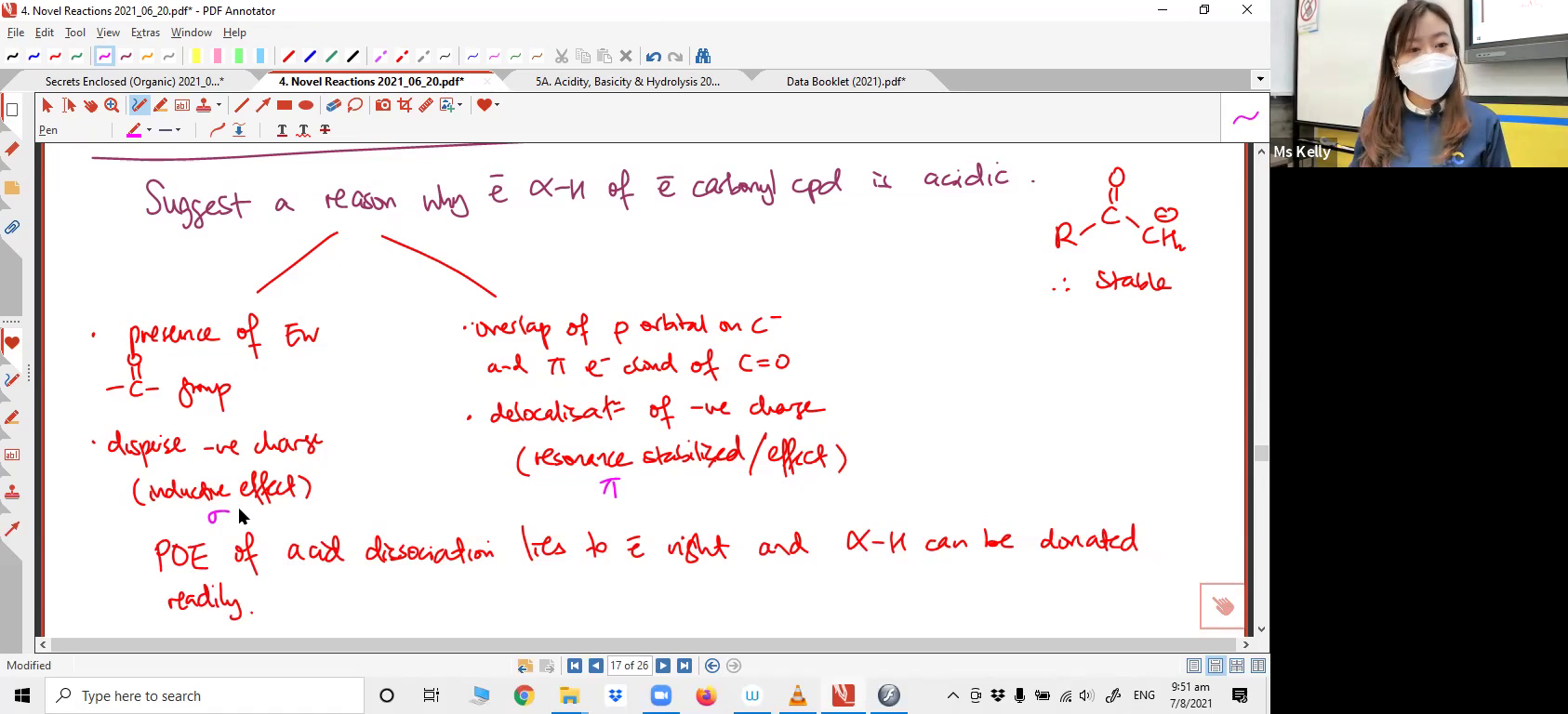
Drawing the mechanism. Suggesting reactant from applying the reaction involving epoxide.6. Tautomerism
Drawing the mechanism. Suggesting a reason for the acidic nature of a particular compound.7. Aryl Halides undergoing Nucleophilic Substitution
Understanding how aryl halides can undergo a limited number of Nucleophilic Substitution reactions with strong nucleophiles. Suggesting the reaction mechanism of another compound based on given info about aryl halide nucleophilic substitution.8. Formation of esters from carboxylic acids and diazomethane
Drawing the mechanism about an alternative way to form esters. Protonation of diazomethane by carboxylic acid to yield two different ions, and reacting with carboxylate ion to form N2.9. H2 Chem Paper 1 Deduction Skills
A compilation of MCQ questions about deducing the final product of unknown reactions. Test your observation and deduction skills here!10. Unknown Mechanisms of Elimination Reactions
Just like SN2 and SN1 mechanisms, the E2 and E1 mechanisms for Elimination reactions also exist! (Just that you don't study them in A-Levels)11. Converting protonated molecules to cyclic molecules
Movement of electrons from the C=C double bond to expel a H2O molecule, forming a cyclic compound. Are you able to draw out this mechanism?12. Intermolecular and Intramolecular Electrophilic Substitution
Limonene can be transformed from a non-aromatic compound into an aromatic compound via p-menthane! Can you apply the Electrophilic Substitution mechanism to a reaction you've never heard of?13. Nucleophilic Acyl Substitution Mechanism involving Anhydrides
Do you have a strong understanding of the nucleophilic substitution mechanism? Try applying it to a reaction that's outside your syllabus!14. Grignard Reagent Grade A Question (Part 1)
Grignard Reagent Data Response (Part 1)15. Grignard Reagent Grade A Question (Part 2)
Grignard Reagent Data Response (Part 2)